 
19 July 2022 (primary course – for children 6 months and over) (6 month to 5 year formulation)ģ0 August 2022 (booster – for 18 years and over) (bivalent original/Omicron BA.1 formulation).17 February 2022 (primary course - for 6 years and over) (half the adult dose).7 December 2021 (booster – for 18 years and over).3 September 2021 (primary course - for 12 years and over).9 August 2021 (primary course - for 18 years and over).The Therapeutic Good Administration (TGA) provisionally approved it for use in Australia on: Moderna is approved and available for use as a booster in people aged 18 years and over. Spikevax (Moderna) is approved and available for use as a primary course in people aged 6 months and over. Both vaccines can cause various adverse effects, but these reactions are reported to be less frequent in the Pfizer/BioNTech vaccine compared to the Moderna COVID-19 vaccine however, the Moderna vaccine compared to the Pfizer vaccine is easier to transport and store because it is less temperature sensitive.Information on this page has been superseded by new ATAGI advice on the preferential use of bivalent COVID-19 vaccines for primary course and will be updated shortly. These vaccines can protect recipients from a SARS-CoV- 2 infection by formation of antibodies and provide immunity against a SARS-CoV-2 infection. 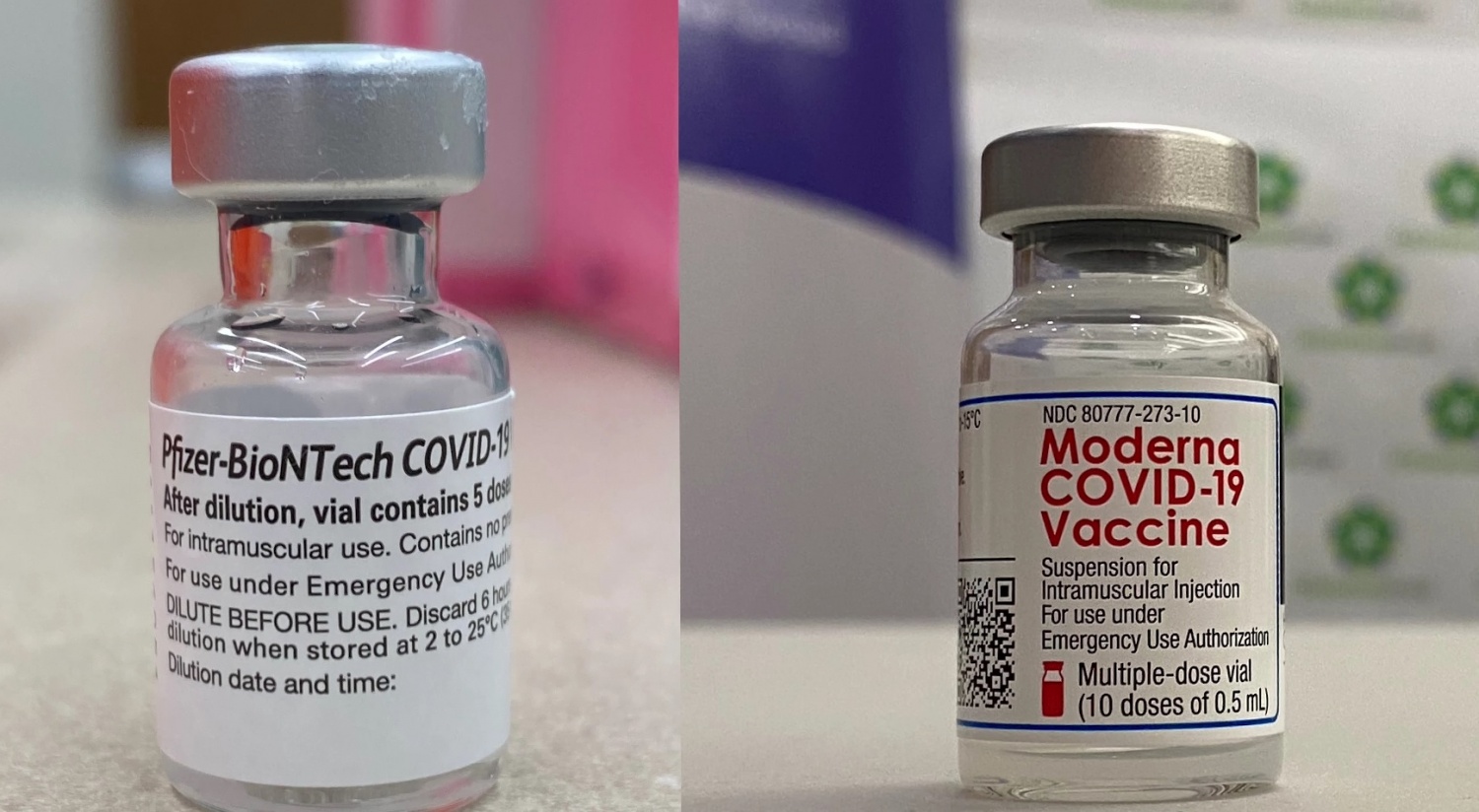
The FDA has granted emergency use authorization for the Pfizer/BioNTech and Moderna COVID-19 vaccines. The occurrence of adverse effects is reported to be lower in the Pfizer/BioNTech vaccine compared to the Moderna vaccine however, the Moderna vaccine compared to the Pfizer vaccine is easier to transport and store because it is less temperature sensitive. The COVID-19 vaccines can cause mild adverse effects after the first or second doses, including pain, redness or swelling at the site of vaccine shot, fever, fatigue, headache, muscle pain, nausea, vomiting, itching, chills, and joint pain, and can also rarely cause anaphylactic shock. However, some associated allergic symptoms have been reported for both vaccines. It provides immunogenicity for at least 119 days after the first vaccination and is 94.5% effective in preventing the SARS-CoV-2 infection. However, Moderna Vaccine has been recommended to people 18 years of age and older, with a dose of 50 μg (0.5 mL) at a cost of $32-37. It provides immunogenicity for at least 119 days after the first vaccination and is 95% effective in preventing the SARS-COV-2 infection. Pfizer/BioNTech Vaccine has been recommended to people 16 years of age and older, with a dose of 30 μg (0.3 m) at a cost of $19.50. The descriptive information was recorded, and we eventually included 12 documents including research articles, clinical trials, and websites to record the required information.īased on the currently available literature, both vaccines are beneficial to provide immunity against SARS-CoV-2 infection. 
The key terms used were: Coronavirus, SARS-COV-2, COVID-19 pandemic, vaccines, Pfizer/BioNTech vaccine, Moderna vaccine, pharmacology, benefits, allergic responses, indications, contraindications, and adverse effects. The required documents and information were collected from the relevant databases, including Web of Science (Clarivate Analytics), PubMed, EMBASE, World Health Organization (WHO), Food and Drug Authorities (FDA) USA, Local Ministries, Health Institutes, and Google Scholar. This article compares the pharmacology, indications, contraindications, and adverse effects of the Pfizer/BioNTech and Moderna vaccines. Many people have a history of a significant allergic reaction to a specific food, medicine, or vaccine hence, people all over the world have great concerns about these two authorized vaccines. The US Food and Drug Administration, (FDA) has granted emergency use authorization for treatment with the Pfizer/BioNTech and Moderna COVID-19 vaccines. The "Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)" disease has caused a worldwide challenging and threatening pandemic (COVID-19), with huge health and economic losses. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
